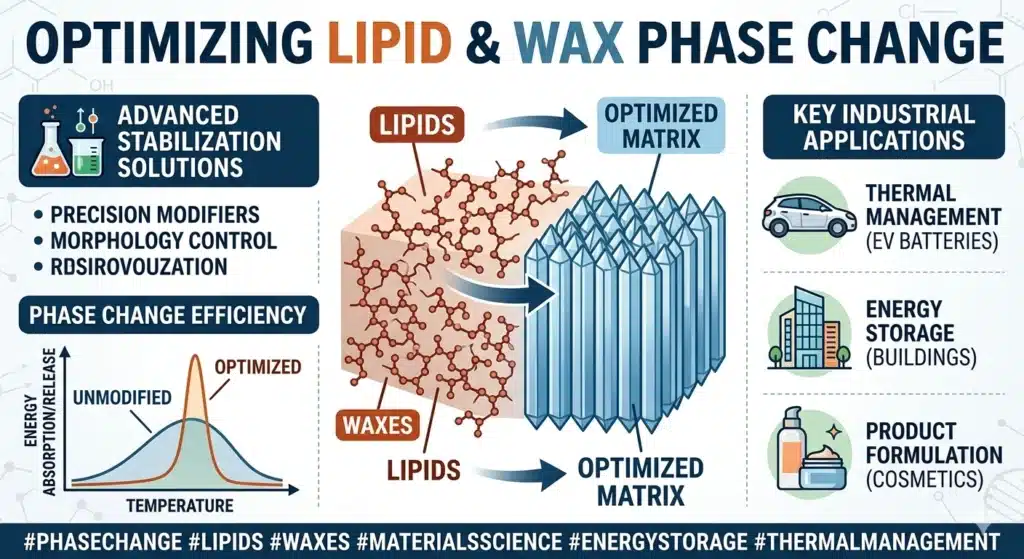
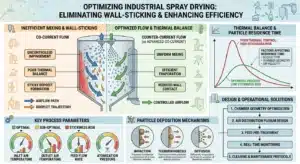
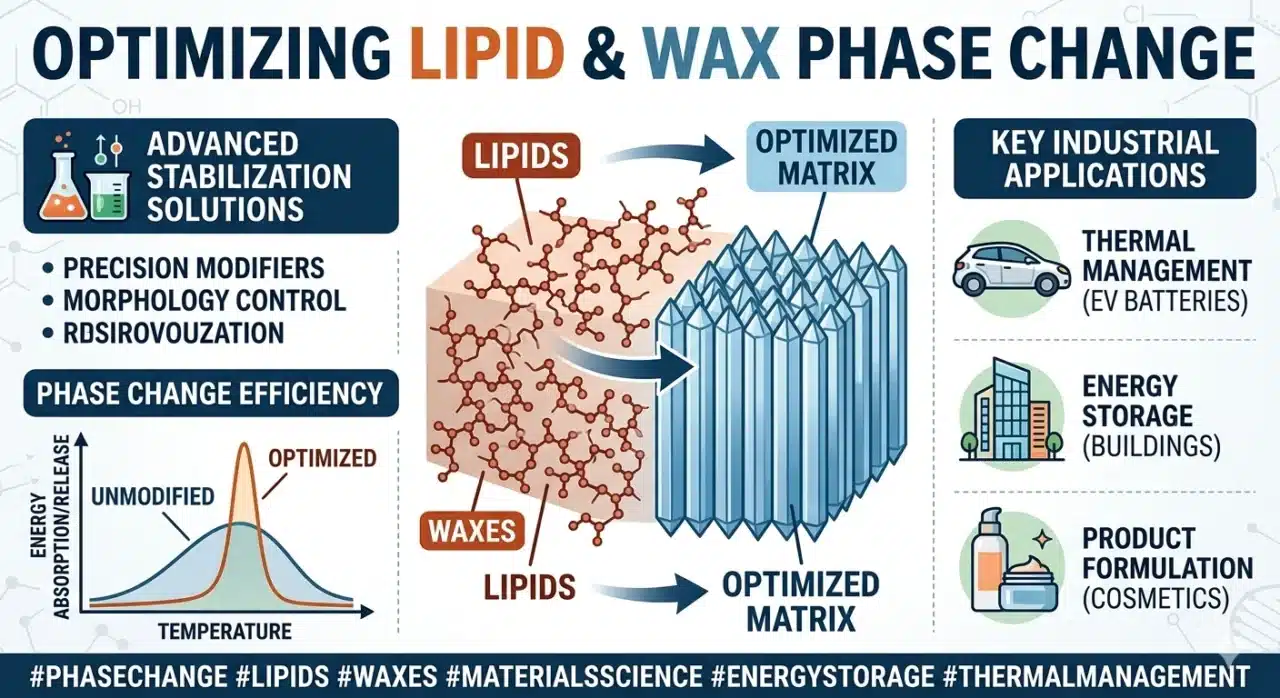
Spray congealing, also known as spray cooling or spray chilling, is a high-precision, solvent-free particle engineering technology that transforms molten lipophilic materials into well-defined solid microparticles through a rapid and controlled phase transition from liquid to solid. Unlike traditional spray drying, which involves the endothermic removal of moisture, spray congealing is an exothermic process where atomized droplets of a melt—comprised of fats, waxes, or hydrogenated oils—solidify upon contact with a cooling gas. SINOTHERMO addresses the fundamental challenge of lipid polymorphism, where the carrier material can transition between unstable ![]() Dan
Dan ![]() stable crystal forms, by implementing “surgical” precision in thermal gradient management. This approach utilizes a meticulously calibrated cooling curve to govern the three critical stages of particle formation: liquid sensible heat removal, the release of latent heat during crystallization, and post-solidification sub-cooling. By maintaining strict control over atomization pressure, melt viscosity, and chamber thermodynamics, SINOTHERMO ensures near 100% encapsulation efficiency and long-term shelf stability, effectively preventing the premature expulsion of active ingredients common in less precise cooling systems. This technology represents the pinnacle of B2B solutions for masking bitter tastes, achieving controlled release, and protecting sensitive bioactives in the pharmaceutical, nutraceutical, and specialty food sectors.
stable crystal forms, by implementing “surgical” precision in thermal gradient management. This approach utilizes a meticulously calibrated cooling curve to govern the three critical stages of particle formation: liquid sensible heat removal, the release of latent heat during crystallization, and post-solidification sub-cooling. By maintaining strict control over atomization pressure, melt viscosity, and chamber thermodynamics, SINOTHERMO ensures near 100% encapsulation efficiency and long-term shelf stability, effectively preventing the premature expulsion of active ingredients common in less precise cooling systems. This technology represents the pinnacle of B2B solutions for masking bitter tastes, achieving controlled release, and protecting sensitive bioactives in the pharmaceutical, nutraceutical, and specialty food sectors.
The successful manufacture of lipid-based microparticles relies on a deep understanding of the thermodynamic behavior of the carrier matrix during cooling. Lipophilic substances, particularly triglycerides and waxes, do not exhibit a simple, singular freezing point. Instead, they undergo complex crystallization processes influenced by their molecular structure and the rate of heat removal.
Categorization by Thermal Profile
The industrial nomenclature for these processes is primarily determined by the melting point (![]() ) of the carrier material and the corresponding environmental temperature required to induce solidification. While the terms are often used interchangeably, professional engineering standards define them based on specific temperature ranges.
) of the carrier material and the corresponding environmental temperature required to induce solidification. While the terms are often used interchangeably, professional engineering standards define them based on specific temperature ranges.
| Process Terminology | Melting Point Range (°c) | Typical Applications | Carrier Materials |
| Spray Chilling | 32 °c to 42°c | Food ingredients, enzymes | Fractionated fats, hydrogenated palm oil |
| Spray Cooling | 45°c to 122°c | Controlled release, taste masking | Stearic acid, carnauba wax, paraffins |
| Spray Congealing | General Term | Broad industrial use | Thermoplastic polymers, high-melt lipids |
These materials are selected for their biocompatibility, GRAS (Generally Recognized as Safe) status, and their ability to protect sensitive active pharmaceutical ingredients (APIs) or nutritional components from environmental degradation. The choice of carrier directly impacts the release kinetics, as lipophilic matrices typically provide sustained release through erosion or diffusion mechanisms.
Polymorphism is the ability of a substance to exist in more than one crystalline form. For lipids, these polymorphs are characterized by different molecular packing arrangements, which in turn dictate their physical properties such as melting point, density, and stability. The three primary forms are ![]() (alpha),
(alpha), ![]() (beta-prime), and (beta).The stability and energy levels of these forms follow a hierarchical order:
(beta-prime), and (beta).The stability and energy levels of these forms follow a hierarchical order:
-
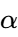 form: The least stable, formed by rapid cooling. It has a hexagonal sub-cell structure and the lowest melting point.
form: The least stable, formed by rapid cooling. It has a hexagonal sub-cell structure and the lowest melting point. -
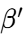 form: An intermediate, metastable form with an orthorhombic sub-cell. It is often preferred in food applications for its texture.
form: An intermediate, metastable form with an orthorhombic sub-cell. It is often preferred in food applications for its texture. -
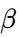 form: The most stable, characterized by a triclinic sub-cell and the highest density. It is the form lipids naturally transition toward over time.
form: The most stable, characterized by a triclinic sub-cell and the highest density. It is the form lipids naturally transition toward over time.
In a spray congealing environment, the rapid cooling of atomized droplets almost always leads to the initial formation of the unstable ![]() polymorph. The challenge for SINOTHERMO is to manage the subsequent transition. If the
polymorph. The challenge for SINOTHERMO is to manage the subsequent transition. If the ![]() →
→![]() transition occurs uncontrollably during storage, the tightening of the crystal lattice can “squeeze out” the encapsulated drug, leading to a phenomenon known as drug expulsion.
transition occurs uncontrollably during storage, the tightening of the crystal lattice can “squeeze out” the encapsulated drug, leading to a phenomenon known as drug expulsion.
To achieve “surgical” precision in particle engineering, SINOTHERMO systems focus on the management of the thermal gradient—the temperature difference between the core of the droplet and the cooling medium. This gradient determines the rate of heat transfer and, consequently, the crystallization kinetics.
The Solidification Curve
The transition from a molten droplet to a solid particle occurs along a specific solidification curve, which can be divided into three distinct thermodynamic stages:
- Stage 1: Liquid Sensible Heat Removal
As the droplets leave the atomizer at a temperature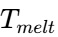 (typically 10°cabove the melting point), they immediately begin to lose heat to the cold air. The rate of cooling is governed by the formula:
(typically 10°cabove the melting point), they immediately begin to lose heat to the cold air. The rate of cooling is governed by the formula:
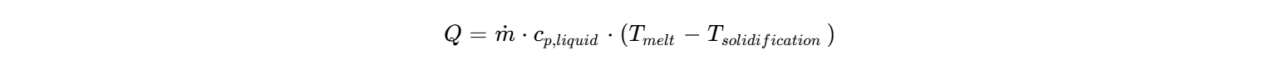
Where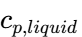 is the specific heat of the melt. SINOTHERMO ensures that the atomizing air is also heated to prevent premature solidification within the nozzle, which could lead to clogging.
is the specific heat of the melt. SINOTHERMO ensures that the atomizing air is also heated to prevent premature solidification within the nozzle, which could lead to clogging. - Stage 2: Latent Heat of Crystallization Once the droplet reaches its solidification temperature, the temperature remains relatively constant despite continued heat removal. This “plateau” is where the latent heat of fusion (
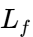 ) is released as the molecules organize into a crystalline lattice. This is the most critical stage for managing polymorphism. If heat is removed too aggressively, the lipid may enter a “glassy” state or form highly defective
) is released as the molecules organize into a crystalline lattice. This is the most critical stage for managing polymorphism. If heat is removed too aggressively, the lipid may enter a “glassy” state or form highly defective 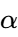 crystals.
crystals. - Stage 3: Solid-State Sub-cooling After the particle has fully solidified, it continues to cool until it reaches the exit temperature of the chamber. This stage is vital for ensuring the particles are below their “sticky point” temperature (
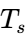 ) before they come into contact with the collection surfaces.
) before they come into contact with the collection surfaces.
The efficiency of heat transfer is highly dependent on the surface-area-to-volume ratio of the droplets. Smaller droplets cool faster, which can be beneficial for throughput but risky for crystal stability. SINOTHERMO utilizes advanced centrifugal atomizers and pressure nozzles to achieve uniform droplet size distributions.Viscosity management is paramount. Feed viscosity is influenced by the solids content, the temperature of the melt, and the presence of additives such as surfactants. Research has shown that maintaining a viscosity below 500 mPas is ideal for atomization. SINOTHERMO’s heated feed systems and insulated pipelines maintain the necessary fluid characteristics throughout the process, ensuring that the energy required for atomization is minimized and the resulting particles are dense and spherical.
In the B2B landscape, equipment must not only perform but also comply with rigorous international standards. SINOTHERMO systems are engineered to meet the stringent requirements of the FDA, GMP, and EU-GMP Annex 1.
Clean-in-Place (CIP) and Material Selection
The processing of lipids presents a unique cleaning challenge. Fats and waxes can become highly adhesive if they cool on the internal surfaces of the machine. To mitigate this, the ZLPG Extract Spray Dryer model from SINOTHERMO incorporates a cylinder cooling system that maintains wall temperatures below 80°c , preventing the lipid from melting and sticking or degrading.For thorough sanitation, SINOTHERMO employs advanced CIP systems featuring:
- Retractable Spray Nozzles: These nozzles remain flush with the wall during production to avoid process disturbance and extend into the chamber during cleaning.
- 360-degree Rotating Spray Balls: Ensuring every square inch of the chamber, including difficult-to-reach areas behind baffles and nozzles, is reached by the cleaning solution.
- Validated Cleaning Parameters: SINOTHERMO provides protocols for alkaline washes (to saponify lipids) and acid washes (to remove mineral deposits), with typical cycle times ranging from 65 to 115 minutes.
| Cleaning Component | Material | Function |
| Chamber Walls | SUS316L | High-grade corrosion resistance and smoothness (Ra < 0.4 |
| Gaskets | EPDM / PTFE | FDA-approved food-grade compatibility |
| Spray Balls | SUS316L | Dynamic rotation for impingement cleaning |
Recovery Systems and Yield Optimization
Maximizing the recovery of high-value active ingredients is a primary objective. In spray congealing, the yield is typically very high—often 95-100%—because there is no solvent loss. However, fine particles can still be lost in the exhaust stream.
SINOTHERMO utilizes high-efficiency cyclone separators and pulse-jet bag filters to capture these fines. The design of these recovery systems is optimized to prevent “caking,” which occurs when particles are stored above their glass transition temperature (![]() ). Furthermore, the integration of pneumatic hammers or air brushes ensures that any powder adhering to the walls is vibrated or swept into the collection cone, maintaining continuous operation and consistent batch yields.
). Furthermore, the integration of pneumatic hammers or air brushes ensures that any powder adhering to the walls is vibrated or swept into the collection cone, maintaining continuous operation and consistent batch yields.
A critical comparison for B2B decision-makers is the choice between spray congealing and alternative size-reduction or encapsulation methods, such as cryogenic grinding.
The Limitations of Cryogenic Grinding
Cryogenic grinding involves cooling a material below its glass transition temperature using liquid nitrogen and then mechanically milling it. While effective for some polymers, it has several drawbacks for lipids:
- Irregular Morphology: Grinding produces jagged, non-spherical particles, which have poor flowability and lower bulk density.
- Surface Exposure: The mechanical fracturing of the lipid matrix can expose the encapsulated API on the surface, defeating the purpose of taste masking or protection.
- Pemrosesan Batch: It is often a multi-step, batch process, whereas spray congealing is inherently continuous.The Advantages of Continuous Congealing
Spray congealing is the “solvent-free equivalent of spray drying”. It allows for the production of dense, spherical, free-flowing microparticles in a single step. The lack of an evaporation step means that the particles are non-porous, providing a superior barrier against moisture and oxygen.From an energy perspective, while spray drying is an energy-intensive endothermic process (typically consuming 4.3 MW for a 90,000 kg/h system), spray congealing requires energy primarily for melt heating and cooling-gas refrigeration. SINOTHERMO’s advanced heat recovery systems can capture up to 35% of the energy from the exhaust stream, significantly reducing the carbon footprint and operational costs of the facility.
The application of SINOTHERMO’s technology extends beyond simple lipid matrices into complex delivery systems.
Enhancing Bioavailability through Solid Dispersions
For poorly water-soluble drugs (BCS Class II and IV), spray congealing can be used to create solid dispersions. By dispersing the drug at a molecular level within a hydrophilic lipid matrix, the surface area for dissolution is dramatically increased. Studies on carbamazepine and sodium diclofenac have shown increases in dissolution rates of up to 2.7 times compared to the pure API.
A novel approach involves using a water-in-oil emulsion as the feed material. By dissolving a hydrophilic drug (like metoclopramide hydrochloride or insulin) in an internal aqueous phase and then congealing the outer lipid phase, a “multicore-shell” particle is created. This structure provides a unique release profile, combining the benefits of a hydrophobic lipid barrier with the stability of a solution-based internal core.
The addition of lipid modifiers such as stearic acid or cetyl alcohol can be used to tune the viscosity and the final crystal structure. For instance, the addition of ![]() of specific liquid lipids has been shown to accelerate the transition from the metastable
of specific liquid lipids has been shown to accelerate the transition from the metastable ![]() form to the stable
form to the stable ![]() form. This “pre-stabilization” is an area where SINOTHERMO’s process engineers provide immense value, assisting clients in selecting the right excipient blend to ensure that the product is stable upon exit from the machine.
form. This “pre-stabilization” is an area where SINOTHERMO’s process engineers provide immense value, assisting clients in selecting the right excipient blend to ensure that the product is stable upon exit from the machine.
Conclusion: The SINOTHERMO Precision Advantage
Mastering phase change in lipids and waxes is not merely about cooling a melt; it is about the sophisticated orchestration of thermodynamic variables to achieve molecular stability. SINOTHERMO’s surgical precision in thermal gradient management solves the industry’s most pressing challenges—polymorphic instability, drug expulsion, and low recovery rates.
By providing a continuous, solvent-free, and high-yield manufacturing platform, SINOTHERMO enables B2B partners to produce superior microencapsulated products that meet the highest standards of the pharmaceutical and food industries. Whether the goal is to mask the taste of a bitter antibiotic, protect a delicate enzyme, or control the release of a potent drug, SINOTHERMO’s advanced spray cooling solutions offer a robust, efficient, and data-driven path to success.